As we transition from Biology to Chemistry, we are moving even smaller still, from looking at things like our DNA to try to understand the bonds that hold atoms together. We will learn about different types of chemical reactions, the Law of Conservation of Mass, and along the way will hopefully enjoy some labs and some great demos.
Lesson #1. Atomic Structure
While this should be review, we will start at the most basic level: what is an atom and what is it made up of. In this class we will meet a group of elements, and do some practice learning about their basic structure. Attached is the powerpoint for the class, as well as a basic worksheet.
It is important to recognize how the elements were created in the first place. This also ties in very well to the Astronomy unit and stars. Here, in this short video, Brian Cox very nicely explains how exactly is was that our more complex elements were created by the death of stars.
|
As we continue with our Big History Project, we realize that the death of stars is what gave and continues to give birth to every element in our world. All life is derived from these stars, and with the creation of more complex elements, the Universe was able to start building more complex structures.
| ||||||||||||
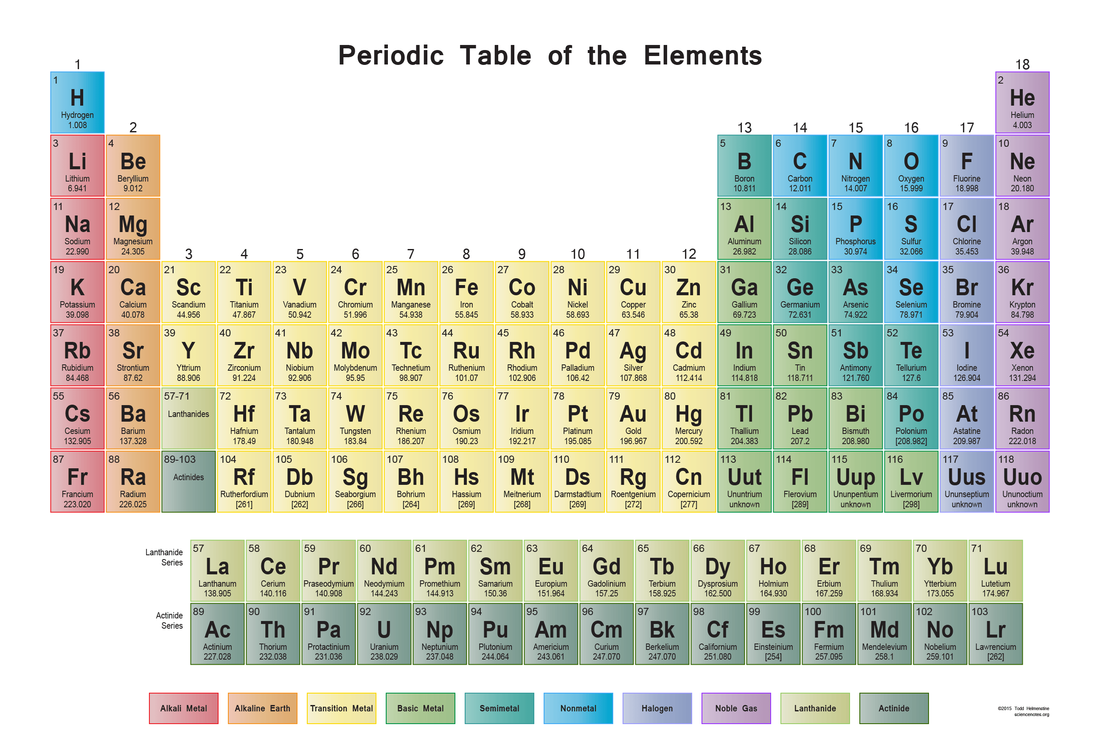
Lesson. #2. Periodic Table of Elements
Although this should be a review of Grade 9 science, we will spend the day re-visiting the table of elements, and refreshing our memory on all the information contained within. Looking at metals/non/metals/halogens, etc...as well as what the groups and periods are telling us, will provide us with the foundation for moving forward in this unit.
|
|
|
| ||||||||||||||||||
Lesson #3. Bohr Diagrams
Having an understanding of the arrangement of electrons for each atom in an element is a crucial piece of knowledge. It allows us to better understand and predict the types of reactions that we will be getting to see soon. Attached are notes and powerpoint slides.
A boring, but clear video on how to draw Bohr diagrams, if you missed the class.
|
| ||||||||||||||||||
Lesson #4. Ionic Bonding
We start to investigate simple compounds: metal + non metal, and how they transfer electrons to form stable ionic bonds. This involves a whole lot of practice of going from name to formula and from formula to name. Notes, as well as worksheet and answer keys, are included.
Clear and simple video that explains how to name basic formulas for simple ionic compounds.
|
| ||||||||||||||||||
Lesson #5. Multivalents
If only it were so easy. Many elements can only have 1 ionic charge. These have a single combining capacity. But others have multiple combining capacities. As in, they can have different ionic charges. These are called multivalents, and require their own practice.
While this is not edge of your seat excitement, this video quite nicely explains how to go about naming and writing formulas for mutivalent metals. Helpful as a reminder, or if you missed the class.
|
| ||||||
Lesson #6. Polyatomic Compounds
One last tricky naming game: polyatomics. No, you won't have to ever memorize all these polyatomics, but you do have to know how to go from name to formula and from formula to name.
|
| ||||||||||||||||||
Lesson #7. Covalent Bonding
Another way that compounds are formed is through covalent bonding. In this case, electrons are shared rather than transferred. This is often the case between non-metals and other non-metals.
|
| ||||||||||||||||||
Lesson #8. Balancing Chemical Equations
We now start to put all of our learning around ionic and covalent compounds together and learn to balance complex chemical equations. There is no magic to this, it is simply about practicing and practicing until one feels confident in their ability to do this. There are several worksheets and answer keys posted below, as well as a little video explaining how to balance a formula, if you missed class.
|
|
| ||||||||||||||||||
Lesson #9. Endothermic and Exothermic Reactions
This is a lab that explains the concept of endo- and exo- thermic reactions. One absorbs energy (heat) and makes its surroundings colder, and the other releases heat into its surroundings and makes them hotter. A simple concept, but an important one to always keep in mind.
|
I have attached a brief little powerpoint, as well as a more in-depth video that really explains all the essentials of the concept.
|
| ||||||
Lesson#10. Types of Reaction
It is important to understand the various types of reactions that one sees in a chemistry classroom. As one's skills progress, being able to predict a reaction is the next level of understanding. Initially, we look at the major types of reactions and the demos and labs we have used along the way as examples of them.
|
| ||||||||||||||||||
Lesson #11. Acids and Bases
We seek to better understand the everyday acids and bases that surround us in our homes, and in the foods and products we consume everyday. I have included a very simple powerpoint and some notes, however, most of the learning for this topic takes places in a lab setting.
| 7a_-_acid_and_bases_ppt.ppt | |
| File Size: | 4048 kb |
| File Type: | ppt |
|
As our universe continue to add complexity over time, through ionic and covalent bonding, our Earth and Solar System are formed. This brings us to the 4th Threshold in our Big History Project.
|
|
Mini-Research Project
In celebration of Earth Week here at HSS, and after watching the documentary "Before the Flood", students will conduct a short research assignment, entitled "How Chemistry Can Help Save the World." The assignment link is below.
| using_chemistry_to_save_the_world.docx | |
| File Size: | 14 kb |
| File Type: | docx |
REVIEW
Time for some review. Below is my review sheets and the answer key.
| antony_practice_exam_answer_key.doc | |
| File Size: | 104 kb |
| File Type: | doc |